GxP Compliance & Data Integrity
We have extensive experience with comprehensive testing and formal validation of information technology systems in a regulated environment in compliance with guidelines such as GxP, 21 CFR Part 11, EU Annex 11, GAMP 5, MHRA, EMEA, HIPAA, and SOX including validation plans, protocol development, UAT, IQ/OQ/PQ, and traceability matrices.
CSV (Computer System Validation) - traditional approach
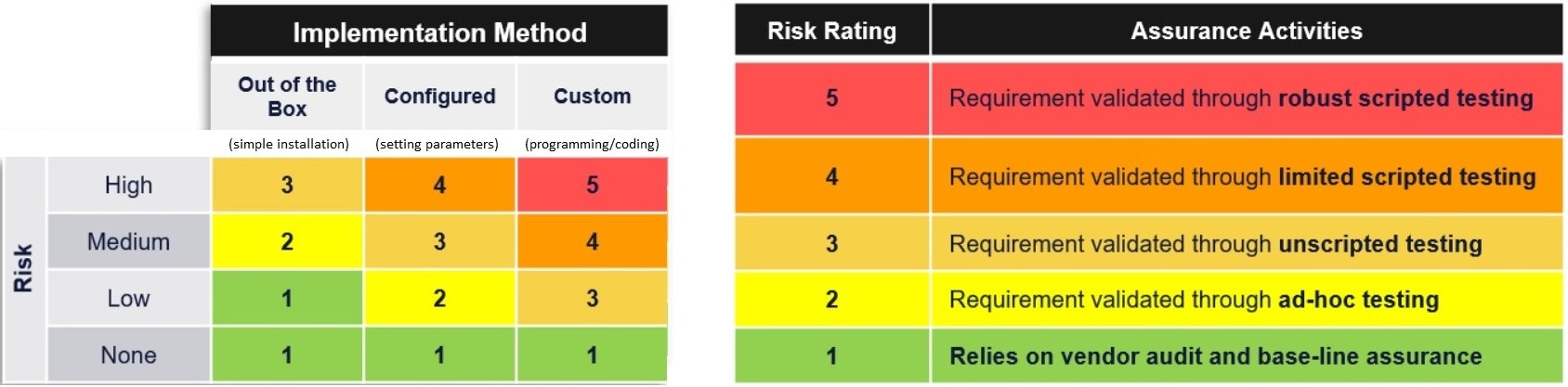
CSA (Computer Software Assurance) - new guidance from FDA. A risk-based approach for establishing and maintaining confidence that software is fit for its intended use. Least-burdensome approach for testing and documentation, commensurate with risk.
Risk-based testing includes:
• leveraging vendor/supplier testing
• ad-hoc testing
• unscripted testing
• limited scripted testing
• robust scripted testing
Data Integrity (ALCOA++)
With the growing emergence of AI, it is vital to sharpen focus on data quality and data integrity, building on fundamentals to further enhance enterprise data quality and increase data integrity, both being absolutely essential to improving AI capabilities and maximizing the leverage it can provide. This requires implementing scalable data platforms, robust metadata standards, and high-performance computing environments designed and optimized for AI/ML workflows and automation. Working to ensure that data is centralized, visible, accessible, interoperable, well-governed, appropriately safeguarded and fully aligned and compliant with all necessary data integrity requirements and other mandates per regulatory, sponsor, and institutional requirements, along with having integrated platforms that enforce standardization will enhance analytic capabilities by providing sharper unified views and more sophisticated holistic insights.
Audit Preparation & Inspection Readiness
Ensuring that documentation is complete, comprehensive, consistent, and easily retrievable, training records are current, processes are consistent with SOPs, and quality management is robust with verifiable traceability are all part of maintaining ongoing strong compliance and operational excellence. We can partner with your organization’s cross-functional SMEs to help plan and orchestrate the necessary preparation for and management of audit and inspection prerequisite tasks for readiness, team conduct and best practices during audits and inspections, comprehensive response and effective follow-up, and ultimately to help transform what can be viewed as a burdensome regulatory exercise into a fundamental opportunity for organizational growth by strengthening compliance and improving processes.
Click on these specialties to further explore our other services:
Strategic Guidance
Process Innovation
Technology Implementation